Episode 151: Myeloma Series, Pt. 11 - Role of bispecific T-cell engagers for relapsed/refractory multiple myeloma (2026)
In this week’s episode, we continue our journey through the relapsed/refractory myeloma space, with a focus on the role of bispecific T-cell engagers. Another incredibly important conversation that is so important in the current treatment landscape and one that you don’t want to miss.
If you haven’t done so already, be sure to check out our CAR T episode.
This episode is sponsored by The Lymphoma, Leukemia and Myeloma Congress! To learn more and register for the meeting, click here! Be sure to use our SPECIAL TFOC code TFOC40 to save 40% off registration.
What are Bispecific T-Cell Engagers?
Engineered antibodies that bind two specific targets simultaneously, one arm binds CD3 on cytotoxic T-Cells and the other binds a specific target antigen on myeloma cells. This action brings the two cells close together allowing the T-cell to destroy the myeloma cell.
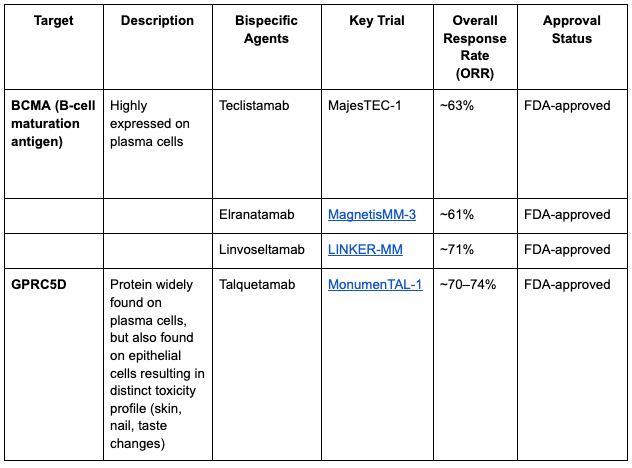
There are four BiTEs currently approved for treating myeloma as of the start of 2026: three of these (teclistamab, elranatamab, and linvoseltamab) target BCMA, while the fourth (talquetamab) targets GPRC5D.
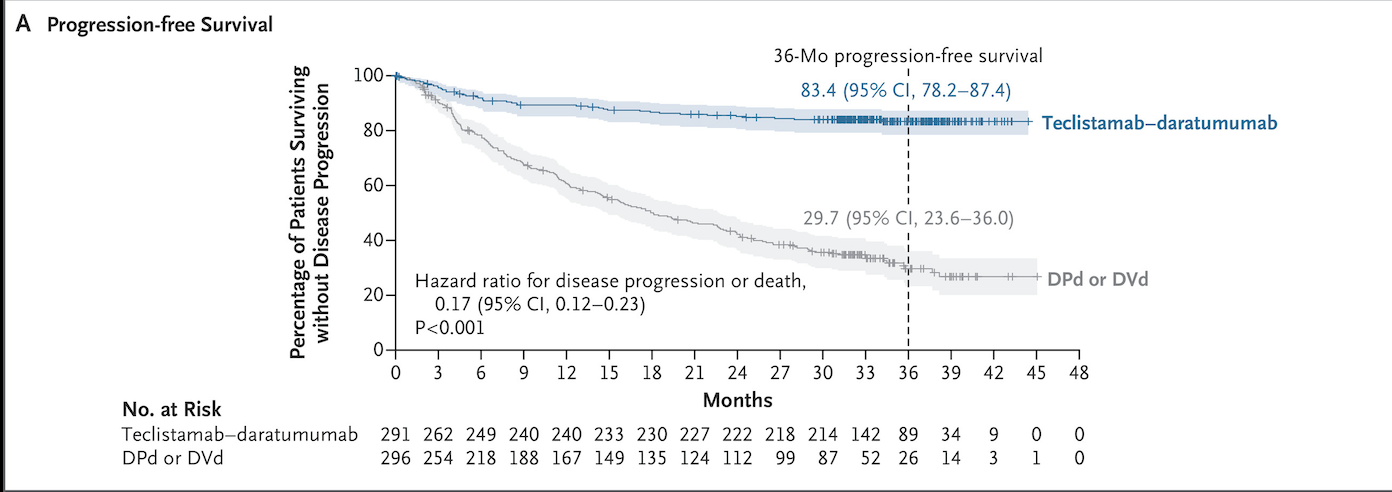
MajesTEC-3: Teclistamab + Daratumumab
MajestTEC-3 was a phase 3 randomized clinical trial, published in December 2025, looking at Teclistamab plus daratumumab (tec-dara) compared to standard of care regimens, including DPd or DVD in relapsed MM. Approved in March 2026.
Image source: Figure 1, MajesTEC-3 Trial (DOI: 10.1056/NEJMoa2514663)
Results
36-month PFS: 83% (tec-dara) vs 30% (SOC)
36-month OS: 83% (tec-dara) vs 65% (SOC)
Demonstrates dramatic improvement in both disease control and survival in earlier-line therapy
Toxicities
CRS and ICANs, though lower than CAR-T therapy
Grade ≥3 infections: 96% (tec-dara) vs 84% (SOC)
Fatal infections 4.6% vs 1.4%, mostly within first 6 months
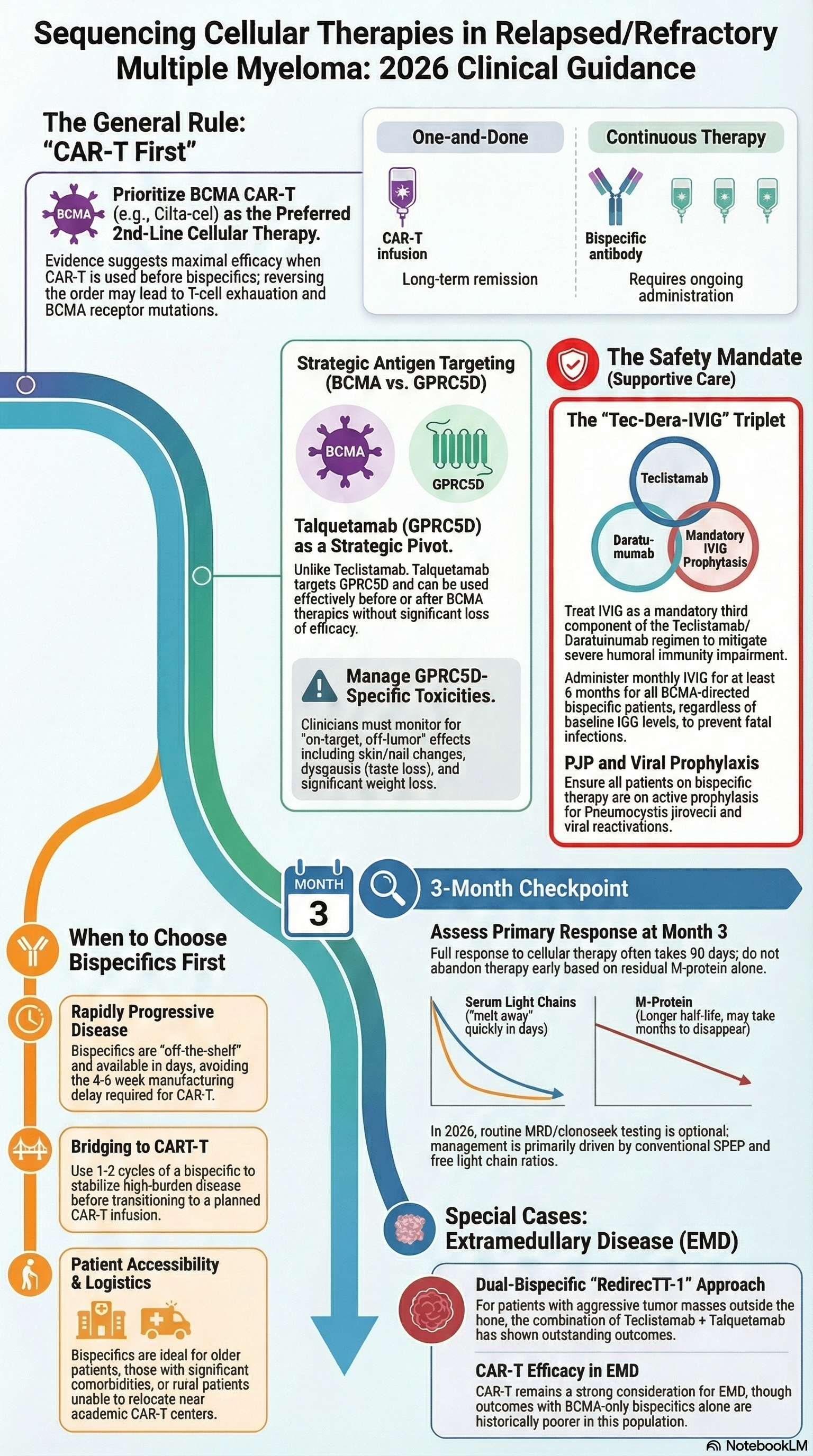
Now we give monthly IVIG as prophylaxis during the first 6 months post BCMA-directed bispecific antibodies
Are there any differences with talquetamab BiTE?
Talquetamab binds GPRC5D which is found on myeloma cells, but also found on epithelial cells, resulting in peculiar side effects. Side effects include rash, fingernail abnormalities, dysgeusia, and anorexia with significant weight loss due to on-target, off-tumor toxicity involving GPRC5D expressing cells elsewhere in the body.
What are the key differences between CAR-T and bispecific therapy?
Bispecific antibodies offer immediate availability and outpatient feasibility, whereas CAR-T provides deeper, potentially more durable responses but requires manufacturing time and inpatient care. Toxicity profiles also differ, with CAR-T associated with higher rates of severe CRS and ICANS.
Does the sequence of when you use CAR-T vs bispecific therapy matter?
Current data suggests using CAR-T followed by bispecific T-cell engagers may yield better outcomes, especially when targeting BCMA. The theory is that this may be due to antigen escape or T-cell exhaustion which is re-amplified by bispecific T-cell engagers. Bispecific T-cell engagers tend to still have a decent response rate when given second, however they do tend to have a shorter PFS in this setting.
The International Myeloma Working Group published consensus guidelines on the sequencing of immunotherapy treatments to help guide as these are rapidly evolving with new agents hitting the market frequently.
Talquetamab represents an important exception, as it targets GPRC5D rather than BCMA. Early data suggest it may retain efficacy after prior BCMA-directed therapies and may not compromise subsequent CAR-T response.
What other factors do you take into consideration when choosing a treatment?
Trajectory of a patient's progression (if rapidly progressing, may need an off the shelf product)
Patient’s underlying comorbidities
Prior lines of therapy
Is there any data for combination bispecific therapy?
The RedirecTT-1 study (NEJM, January 2026) evaluated the combination of talquetamab (anti-GPRC5D) plus teclistamab (anti-BCMA) bispecific antibodies in 90 patients with triple-class–exposed relapsed/refractory multiple myeloma and true extramedullary disease.
Key Results (median follow-up: 12.6 months)
Overall response rate: 79% (95% CI, 69–87%)
12-month PFS: 61%; 12-month OS: 74%
Among responders, 64% maintained response ≥12 months
The dual-antigen targeting approach demonstrated meaningful activity in this high-risk population with historically poor outcomes, though with substantial toxicity consistent with each agent's known profile.
***Our*** summary about how to sequence these therapies:
How we think about sequencing CAR T vs. bispecific T-cell engagers. Infographic created using NotebookLM.
This episode is sponsored by The Lymphoma, Leukemia and Myeloma Congress! To learn more and register for the meeting, click here! Be sure to use our SPECIAL TFOC code TFOC40 to save 40% off registration.
References
Costa LJ, Bahlis NJ, Perrot A, Nooka AK, Lu J, Pawlyn C, Mina R, Caeiro G, Kentos A, Hungria V, Reece D, Niu T, Mylin AK, Hansen CT, Teipel R, Besemer B, Dimopoulos MA, Zamagni E, Yoshihara S, Kim K, Min CK, Geerts P, Van Leeuwen-Segarceanu E, Tyczynska A, Reguera JL, Johansson M, Hansson M, Turgut M, Grey M, Sidana S, Rodriguez-Otero P, Martinez-Lopez J, Hashmi H, Carson R, Kobos R, Sun W, Lantz K, Seifert A, Briseno-Toomey D, O'Rourke L, Rubin M, Vieyra D, Kang L, Mateos MV; MajesTEC-3 Trial Investigators. Teclistamab plus Daratumumab in Relapsed or Refractory Multiple Myeloma. N Engl J Med. 2026 Feb 19;394(8):739-752. doi: 10.1056/NEJMoa2514663. Epub 2025 Dec 9. PMID: 41363801.
Costa, L.J., Banerjee, R., Mian, H. et al. International myeloma working group immunotherapy committee recommendation on sequencing immunotherapy for treatment of multiple myeloma. Leukemia 39, 543–554 (2025). https://doi.org/10.1038/s41375-024-02482-6
Lesokhin, A.M., Tomasson, M.H., Arnulf, B. et al. Elranatamab in relapsed or refractory multiple myeloma: phase 2 MagnetisMM-3 trial results. Nat Med 29, 2259–2267 (2023). https://doi.org/10.1038/s41591-023-02528-9
Naresh Bumma et al. Linvoseltamab for Treatment of Relapsed/Refractory Multiple Myeloma. J Clin Oncol 42, 2702-2712(2024). DOI:10.1200/JCO.24.01008
Chari A, Touzeau C, Schinke C, Minnema MC, Berdeja JG, Oriol A, van de Donk NWCJ, Rodríguez-Otero P, Morillo D, Martinez-Chamorro C, Mateos MV, Costa LJ, Caers J, Rasche L, Krishnan A, Ye JC, Karlin L, Lipe B, Vishwamitra D, Skerget S, Verona R, Ma X, Qin X, Ludlage H, Campagna M, Masterson T, Hilder B, Tolbert J, Renaud T, Goldberg JD, Kane C, Heuck C, San-Miguel J, Moreau P. Safety and activity of talquetamab in patients with relapsed or refractory multiple myeloma (MonumenTAL-1): a multicentre, open-label, phase 1-2 study. Lancet Haematol. 2025 Apr;12(4):e269-e281. doi: 10.1016/S2352-3026(24)00385-5. Epub 2025 Mar 13. PMID: 40090350.
The crew behind the magic:
Show outline: Vivek Patel, Sean Taasan
Production and hosts: Sean Taasan, Vivek Patel, Ronak Mistry
Editing: Resonate Recordings
Shownotes: Megan Connor
Graphics, social media management: Ronak Mistry